Antidote for coumadin
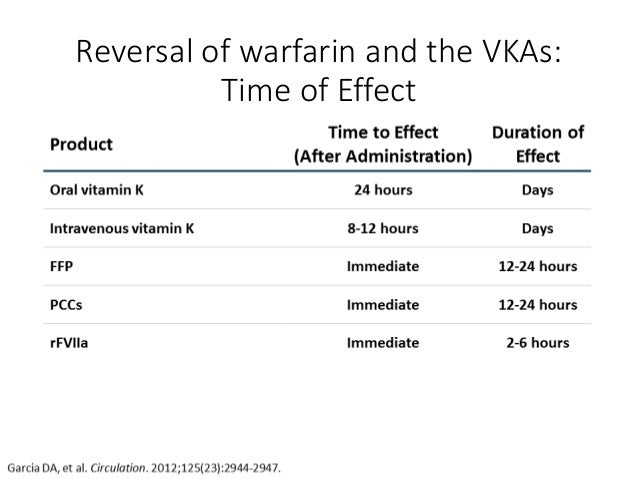
Food and Drug Administration granted accelerated approval for idarucizumab in October 2015 after an interim analysis of the first 90 patients while the RE-VERSE AD (Reversal of Effects of Idarucizumab in Patients on Active Dabigatran) study was ongoing (11). Just as dabigatran became the first DOAC on the market, idarucizumab was first in line for reversal agents. To address this, idarucizumab (Praxbind, Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, Connecticut), a humanized monoclonal antibody fragment, was developed as a reversal agent by specifically binding to dabigatran with high affinity. Up until recently, there was a lack of effective reversal strategies for DOACs, which was indeed troubling given the increased risk of gastrointestinal bleeding with all 4 of the DOACs as demonstrated by a large meta-analysis (10). In this regard, there is still much to learn about renal dosing of the DOACs. Being equipped with a repertoire of alternatives to warfarin and respective reversal agents in the CKD patient cohort, which is commonly afflicted with polypharmacy and is at increased risk for both bleeding and thromboembolic complications, is crucial given the many limitations of vitamin K antagonists with international normalized ratio values often falling outside therapeutic goal as well as the increased risk of vascular calcification and calciphylaxis (9).

Although DOACs have revolutionized the approach to anticoagulation in patients with nonvalvular atrial fibrillation (AF), caution is warranted in patients with CKD and particularly in the elderly, in whom renal function can suddenly decline. However, there have been 2 primary concerns around the use of these DOACs: 1) dosing and safety in patients with chronic kidney disease (CKD) or end-stage renal disease (ESRD) and 2) reversibility. The nomenclature has since been revised and the drugs are now referred to as direct oral anticoagulants (DOACs) to highlight that these drugs are no longer considered “novel.” This spurred a succession of novel oral anticoagulants released into the market, which began with rivaroxaban after the pivotal ROCKET AF (Rivaroxaban Once-Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolist Trial in Atrial Fibrillation) (6), followed by apixaban (ARISTOTLE ) (7), and edoxaban (ENGAGE AF-TIMI 48 ) (8), all direct factor Xa inhibitors. Unlike the vitamin K antagonist mechanism of warfarin, or even the mechanism of heparin which induced anticoagulation by binding to antithrombin III, dabigatran was a truly “novel” drug that directly bound to thrombin to employ its anticoagulant effect. The advent of this new anticoagulant was a major advance given that warfarin was wrought with a panoply of drug–drug interactions, need for frequent monitoring, dietary restrictions, and a narrow therapeutic window increasing the risk of supratherapeutic or subtherapeutic drug levels. Food and Drug Administration approval in 1954. market following the results of the RE-LY (Randomized Evaluation of Long Term Anticoagulant Therapy) trial (5), warfarin was the only oral anticoagulant option since its U.S. However, up until 2010 when dabigatran (Pradaxa) first made its entry into the U.S. We have come a long way from moldy hay, sick cows, bleeding chicks, and rodenticide. Army recruit who ingested rodenticide in 1951 (3), marking the first in-human experience of reversal of anticoagulation. In parallel, although many of the mechanisms remained elusive, vitamin K was identified as a crucial factor in preventing a hemorrhagic disease similar to scurvy in chicks (4), and eventually was used as an antidote in a coumadin overdose by a U.S. This team soon developed several variations of coumarin including number 42, which became known as “warfarin,” first marketed as a rodenticide in 1948 (3). A research team funded by the Wisconsin Research Alumni Foundation (WARF) eventually isolated dicoumarol in 1939 as the culprit agent, which was the result of oxidized coumarin in the moldy sweet clover hay (2). The discovery of modern oral anticoagulation and reversal strategies is a fascinating story of serendipity, which has its beginnings in the 1920s with cattle afflicted with a fatal internal bleeding disorder that was linked to eating moldy hay (1).
